100kwh-hunter
Active member
- Joined
- Mar 2, 2019
- Messages
- 1,374
Update 2: 1st of march 2020
How I gotstarted:
https://secondlifestorage.com/showthread.php?tid=8557
How to build:
https://secondlifestorage.com/showthread.php?tid=8772
This first post will be updated when needed with new test results and cells.
Goal is to find out:
1: there max charging volt.
2: self discharging.
3: what they can hold on power
4: c rate at charge and discharge.
5: behavior when connected in series
6: cycling test.
7: need for under or over charge/voltage protection
8: need for balancing.
9: how much it will take to get them full and how much they will give before empty.
The new cylindrical cells:
Cell one is happy to take 0.3 ah and give 0.3ah with every cycle it is going up, i used 50cc of electrolyte: 40gr of pure ZnBr2 to 100cc distilled water, the max that the water can take without tricks is 44 grams per 100cc(2 M max)
When beginning discharge the cell is at 1.80v after discharge the cell jumps back to 1.77v and i can do a second run
Cell one is going to have its fifth cycle and cell two his first atm.
Cell one holds ~35-40cc electrolyte( i spilled some electrolyte)
Average charge discharge rate 0.200ma
Average capacity 300mah
7 cycles
Cell two holds 50cc electrolyte.
Average charge discharge rate 0.200ma
Average capacity 550mah
Both at 1.80-1.82v
3 cycles
I think those are nice numbers
Max charging voltage seems to be 4.00v, best would be 3.5v, they react exactly as lead acid, but with a unlimited cycle life.
You can feed them 10ah, but they will only take what they want.
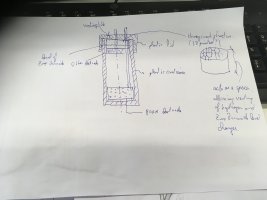
Cell one and two dimensions: 5cm in height and id 57mm.
Self discharging can be to 0.00 volt without damage.
There is so far no need for a bms or balancing.
This message will change from time to time when the numbers are cracked and narrowing down.
For now cell one: 1.80v 50cc of 1.75 M solution will give 350-400mah for the costs of 15% of a new 800 cycle lifeli ion 18650.
Against unlimited cycle life? you do this math...
Will update when i have some more numbers/statistics.
Building of this cell:
https://secondlifestorage.com/showthread.php?tid=8772
Look for things that look similar to a pipe
--------------------------------------------------------------
The cells below are with oasis and closed design, every experiment FAILED...
On power storage, taking and giving some mmmmmmmmmah, lets say femtoamp hours.
There was no flow of the electrolyte and there was to much pressure building up.
One week of charge at 0.02a and burning a led for 10 minutes.....
Above this alina, i will post test results of the new cylindrical cell without oasis, looks really better.....
------------------------------------------------------------
With foam and closed cells...failedon every aspect, the model was wrong
Cell specs as 26-02, with all the same dimensions: 7x5cm and 2 cm thick,
Charge and discharge are done with a 4a dc dc converter, zb2l3 and 3x yellow led (1.6v to 2.1v at 20mahx3=60mah)
Cell 1: No graphite foil and ~60-70% full of electrolyte.
Cell 2: No graphite foil and 100% full
Cell 3: With graphite foil and 100% full
Cell test results:
Cell 1:
Charged several times to 1.82 - 1.85v with 3.5v charge, discharged to 0.00-0.05v several times.
Left on the charger for three days, voltige was 2.15v after been disconnected from the charger for3 hours.
To weak to light up one yellow led by itself.
Cell 2:
Charged severaltimes to 1.82 - 1.85v with 3.5v charge,discharged to 0.00-0.05v several times.
Left on the charger for three days, voltige was 1.82vafter been disconnected from the charger for3 hours.
To weak to light up one yellow led by itself.
Cell 3:
Taking his initial charge:
3 hours at 2.5v, max reached voltige 1.30v: was standing on 1.30vfor over one hour.
Went to 3.6v charging, it takes 0.030ah now.
Adding the graphite foil seems to improve as a collector.
After 4 hours it took 0.200mah!
After adding 3 hours it took 0.125 still loading...
Graphite foil is the way i guess
Cell 4 and 5 failed also...
Not enough mah taking or giving, plus to much presseur.
Various tests:
Connected cell 1 and 2 in series to gain 3.65v and 3 leds in parallel.
Every full cycle, same results: 2.5 hour burning time, cell 1 ended at 0,15v and cell 2 at 1.65v, 3 hour charging in series at 7.2v at 0.025ah
Some dischargetests with cell 1 and 2 after discharge with leds was with a resistor of 5.1 ohm 5 watt to go to 0.00v and charge it back up again to 3.65v at 7.2v charge.
Tested cell 2 allone for charge at 5v and 7v...7v is a no go, the cell had pressure building up inside.
General results up to now:
It seems they must have dubble voltige before they want to take any power into them.
The more electrolyte is in them the more they can storage.
Most tests will be done at the same way at the same cell to get consistent results
With foam and closed cells...failedon every aspect, the model was wrong
If anyone has a idea, thought, comment, testing, behavior or whatsoeverplease do share.
Not that i don't like it, but please give a like if you want me to keep this updated and continue to share my work.
Thanks in advance, reading,understanding and the likes for appreciation for my work....this does not count as hobby anymore
How I gotstarted:
https://secondlifestorage.com/showthread.php?tid=8557
How to build:
https://secondlifestorage.com/showthread.php?tid=8772
This first post will be updated when needed with new test results and cells.
Goal is to find out:
1: there max charging volt.
2: self discharging.
3: what they can hold on power
4: c rate at charge and discharge.
5: behavior when connected in series
6: cycling test.
7: need for under or over charge/voltage protection
8: need for balancing.
9: how much it will take to get them full and how much they will give before empty.
The new cylindrical cells:
Cell one is happy to take 0.3 ah and give 0.3ah with every cycle it is going up, i used 50cc of electrolyte: 40gr of pure ZnBr2 to 100cc distilled water, the max that the water can take without tricks is 44 grams per 100cc(2 M max)
When beginning discharge the cell is at 1.80v after discharge the cell jumps back to 1.77v and i can do a second run
Cell one is going to have its fifth cycle and cell two his first atm.
Cell one holds ~35-40cc electrolyte( i spilled some electrolyte)
Average charge discharge rate 0.200ma
Average capacity 300mah
7 cycles
Cell two holds 50cc electrolyte.
Average charge discharge rate 0.200ma
Average capacity 550mah
Both at 1.80-1.82v
3 cycles
I think those are nice numbers
Max charging voltage seems to be 4.00v, best would be 3.5v, they react exactly as lead acid, but with a unlimited cycle life.
You can feed them 10ah, but they will only take what they want.
Cell one and two dimensions: 5cm in height and id 57mm.
Self discharging can be to 0.00 volt without damage.
There is so far no need for a bms or balancing.
This message will change from time to time when the numbers are cracked and narrowing down.
For now cell one: 1.80v 50cc of 1.75 M solution will give 350-400mah for the costs of 15% of a new 800 cycle lifeli ion 18650.
Against unlimited cycle life? you do this math...
Will update when i have some more numbers/statistics.
Building of this cell:
https://secondlifestorage.com/showthread.php?tid=8772
Look for things that look similar to a pipe
--------------------------------------------------------------
The cells below are with oasis and closed design, every experiment FAILED...
On power storage, taking and giving some mmmmmmmmmah, lets say femtoamp hours.
There was no flow of the electrolyte and there was to much pressure building up.
One week of charge at 0.02a and burning a led for 10 minutes.....
Above this alina, i will post test results of the new cylindrical cell without oasis, looks really better.....
------------------------------------------------------------
With foam and closed cells...failedon every aspect, the model was wrong
Cell specs as 26-02, with all the same dimensions: 7x5cm and 2 cm thick,
Charge and discharge are done with a 4a dc dc converter, zb2l3 and 3x yellow led (1.6v to 2.1v at 20mahx3=60mah)
Cell 1: No graphite foil and ~60-70% full of electrolyte.
Cell 2: No graphite foil and 100% full
Cell 3: With graphite foil and 100% full
Cell test results:
Cell 1:
Charged several times to 1.82 - 1.85v with 3.5v charge, discharged to 0.00-0.05v several times.
Left on the charger for three days, voltige was 2.15v after been disconnected from the charger for3 hours.
To weak to light up one yellow led by itself.
Cell 2:
Charged severaltimes to 1.82 - 1.85v with 3.5v charge,discharged to 0.00-0.05v several times.
Left on the charger for three days, voltige was 1.82vafter been disconnected from the charger for3 hours.
To weak to light up one yellow led by itself.
Cell 3:
Taking his initial charge:
3 hours at 2.5v, max reached voltige 1.30v: was standing on 1.30vfor over one hour.
Went to 3.6v charging, it takes 0.030ah now.
Adding the graphite foil seems to improve as a collector.
After 4 hours it took 0.200mah!
After adding 3 hours it took 0.125 still loading...
Graphite foil is the way i guess
Cell 4 and 5 failed also...
Not enough mah taking or giving, plus to much presseur.
Various tests:
Connected cell 1 and 2 in series to gain 3.65v and 3 leds in parallel.
Every full cycle, same results: 2.5 hour burning time, cell 1 ended at 0,15v and cell 2 at 1.65v, 3 hour charging in series at 7.2v at 0.025ah
Some dischargetests with cell 1 and 2 after discharge with leds was with a resistor of 5.1 ohm 5 watt to go to 0.00v and charge it back up again to 3.65v at 7.2v charge.
Tested cell 2 allone for charge at 5v and 7v...7v is a no go, the cell had pressure building up inside.
General results up to now:
It seems they must have dubble voltige before they want to take any power into them.
The more electrolyte is in them the more they can storage.
Most tests will be done at the same way at the same cell to get consistent results
With foam and closed cells...failedon every aspect, the model was wrong
If anyone has a idea, thought, comment, testing, behavior or whatsoeverplease do share.
Not that i don't like it, but please give a like if you want me to keep this updated and continue to share my work.
Thanks in advance, reading,understanding and the likes for appreciation for my work....this does not count as hobby anymore